The pituitary gland is an important part of the body because it controls several hormonal glands such as adrenals and the thyroid. The gland is a nearly the size of a pea and has two parts referred to as the lobes (Maya & Pressman, 2010). The front part is the anterior pituitary while the back part is the posterior pituitary. The pituitary gland is part of a bony hollow called the pituitary fossa behind the bridge of the nose and below the brain’s base near the optic nerves. Maya and Pressman (2010) add that the pituitary gland is the most important part of the system because it produces hormones controlling the functions of other endocrine glands. The most common problem occurring in the pituitary gland is the development of a benign growth or tumor. The tumor causes the gland to secrete excess hormone, blocks its production, or causes it to malfunction. Other causes of pituitary disorders include traumatic brain injury and pituitary infarction. Although pituitary disorders are able to alter quality of a person’s life significantly, medical or surgical treatment is available where efficient diagnosis is made. The present study investigates the role of imaging modalities in the evaluation of pituitary gland disorders. Particular emphasis is laid on the role of Magnetic Resonance Imaging (MRI) in diagnosing the pituitary gland to find out if it is the most appropriate method for diagnosis.
Anatomy of the pituitary gland
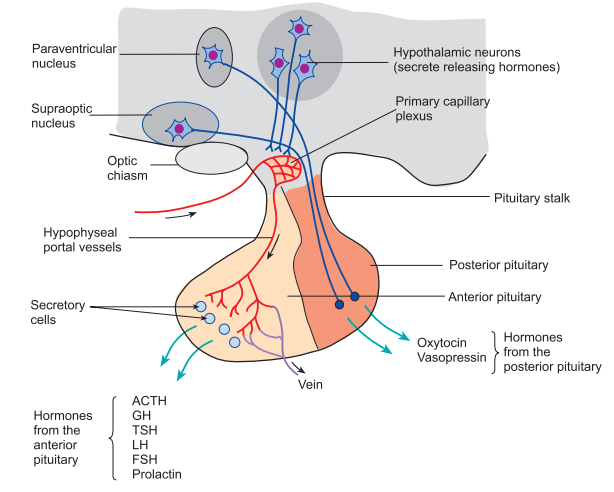
The sellar region of the brain is anatomically complex being bounded by sinus arteroinferiorly, the brainstem posterioly, the dorsum sella, the hypothalamic sellae, the suprasella, and the cavernous sinuses laterally (Drouin, 2010). The hypothelamus consists of a layer of tissue that extends from the anterior to the posterior commissure. The tuber cinereum refers to the inferior ridge of tissue moving from the optic chiasma to mamilliary bodies. Additionally, the hypothalamus consists of multiple nuclei regulating temperature, water balance, and sexual activity as well as drinking behavior. The hypothalamus and the pituitary gland have functional and psychological interlinks between contiguous structures. This is often referred to as the hypothalamic-pituitary axis. The hypothalamus synthesizes oxytocin and vasopressin and transports it to the pituitary gland (Drouin, 2010). The pituitary gland consists of the anterior lobe, which is also known as the adenohypophysis, and makes up 5% of the gland volume. Below is a figure of the pituitary gland.
Figure 1: The Pituitary Gland (Gregory, 2013)
The adenohypophysis includes the pars tuberalis, a vestigial structure, and site for developing cysts, and the pars distalis that makes up most of the intrasellar adenohypophysis (Drouin, 2010). Rathke’s pouch refers to the embryologic precursor of the anterior pituitary gland that regresses at the early stages of developing the fetus. The remaining part of the Rathke’s pouch persists throughout the postnatal life and results in the macroscopic cyst. The adenohypophysis lacks a direct arterial supply, and receives blood from the hypophyseal portal system (Drouin, 2010). This serves as a pathway for releasing hypothalamic hormones. The anterior lobe produces and secretes hormones such as the growth, the adrenocorticotropic, prolactin, thyroid stimulating, luteinizing, and melanocyte-stimulating hormones. When viewed on T1 and T2 weighted images, the anterior lobe is isointense to the cortical brain (Maya & Pressman, 2010).
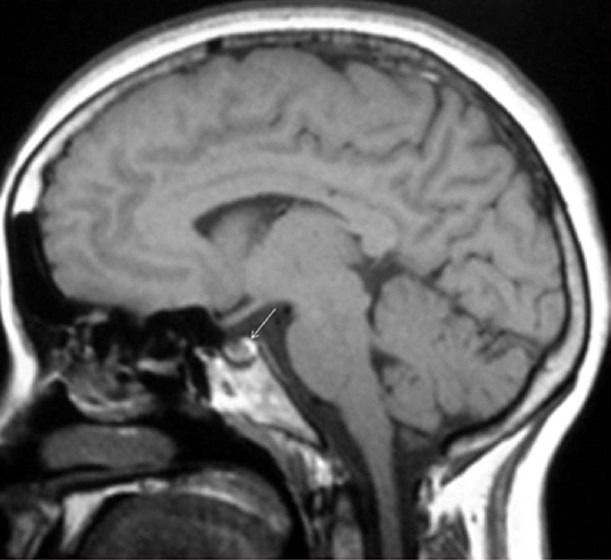
The neurohypophysis includes the pituitary lobe, the supraoptic, and infundibular stock (Maya & Pressman, 2010). The hypothalamus synthesizes oxytocin and vasopressin before transporting along the hypothalamo-hypophyseal tract for storage in the posterior lobe. On T1 weighted images, the posterior lobe is the bright spot as shown below. Neurosecretory vesicles transmit high intensity signals from the posterior pituitary globe. From this, the lack of a high signal link to the central diabetes insipidus or compressed pituitary gland lesions is evident. In relation to dimensions, the pituitary gland has high variations in height with the gland undergoing dramatic changes in size and shape throughout a person’s life (Chaudhary & Bano, 2012). Elster’s rule offers a guide for determining the height in infants and children, which is 6, 8, 10, and 12. The rule considers 8mm for men, 10 mm for women bearing children, 12 mm for women in late pregnancy. Further, the pituitary stock has a normal thickness of 2mm, and does not exceed 4mm or the basilar artillery’s width (Chaudhary & Bano, 2012).
Figure: The arrow points to the bright spot on a sagittal T1 image (Chaudhary & Bano, 2012)
The common pathology of pituitary gland
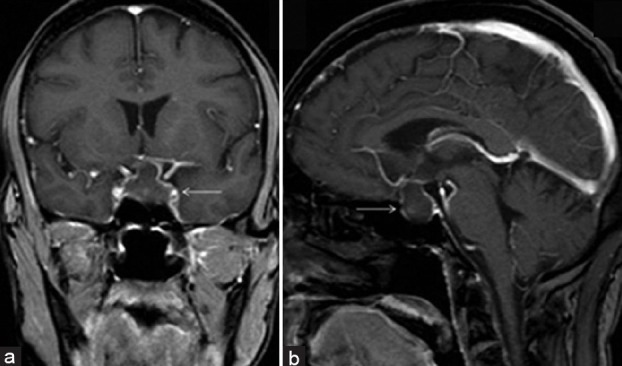
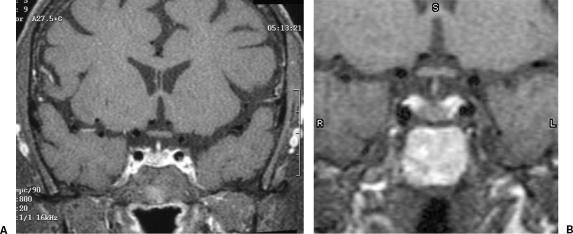
The lacerations in the pituitary gland as well as its sorrounding parts are the main contributors of infection and disease. The effects of the disease are evident from the medical symptoms and indications relating to the endocrinal ssytem, effects of the tumor on the gland or nearby parts, or particulate causes from increased intracranial mass. For instance, pituitary insufficiency alongside a sellar and suprasellar mass lesion suggests a consecratory pituitary tumor that one cannot differentiate clinically from lymphocytic hypophysitis (LH). Where women are presented with sellar mass lesion during pregnancy, the radiologist should suspect lymphocytic hypophysitis (Wisse et al. 2012). However, this can only be ascertained historiologically. An example of an MRI imaging is shown below. Today, MR imaging is considered the best non-invasive diagnostic tool for differentiating lymphocytic hypophysitis from macroadenomas that do not produce any secretions. However, it is important to note that no radiological feature characterizes the disease. The MR features indicating the presence of lymphocytic hypophysitis are symmetric gland enlargements, thickened or enhanced pituitary stalk, or intact sellar floors (Wisse et al. 2012). Other features include a homogenous appearance of the lymphocytic hypophysitis, absence of pituitary bright spot, enhanced adjustments on the pituitary mass.
Figure : Lymphocytic hypophysitis on a patient diagnosed with diabetes insipidus. A is the T1 coronal while b is the T1 sagittal image (Chaudhary & Bano, 2012)
Contrastively, pituitary macroadenomas are asymmetric and homogenous in their appearance. They also have less gadolinium uptake, do not involve stalk, preserve posterior pituitary bright spots, and erode sellar floors. Whereas a thickened pituitary stalk typically shows the presence of a lymphocytic hypophysitis over adonema, enlarged pituitary stalks are found in various diseases such as germinoma, lyphoma, and tuberculosis (Wisse et al. 2012). However, the lack of systemic infections suggets that the presence of hypophysitis. MR imaging increases with the possibility of diagnosing lymphocytic hypophysitis and helps in managing patients with lymphocytic hypophysitis. These patients benefit from medical and not surgical treatment, until a specific antibody for the disease or a character of the MR feature is identified, diagnosis has to rely on historiological study.
Justification for the present study
Currently, the MRI is the most preferred choice of examining sellar and parasellar pathologies given its superior soft tissue contrast (Chaudhary & Bano, 2012). It is also preferred for its multiplanar capability and absence of ionizing radiation. MRIs also offer important information of the link between the gland and the adjacent anatomical structures. This helps in planning medical or surgical surgery. MRI techniques for diagnosing pituitary lesions have increased rapidly from the noncontrast MRI in the 1980s to the contrast enhanced MRIs of the 1990s (Chaudhary & Bano, 2012). Introducing dynamic contrast, enhanced MRI refines the technique when applied to diagnosing pituitary microadenomas. Recently, MR techniques have evolved in evaluating specific cases such as 3D volumetric analysis of pituitary volumes. Others are high imaging 3 Tesla used in evaluating pituitary stock, diffusing weighted imaging, MR spectroscopy, intraoperative MRI and magnetized transfer ratios. To undertsand the development of the MRI as the best means for diagnosing the pituitary gland, the following research objectives and aim will guide the study.
Research aim
- Is Magnetic Resonance Imaging the best means for diagnosing the pituitary gland?
Research Objectives
- What are the best sequences and facts for imaging the pituitary
- What is the role of injection contrast in diagnosing microadenoma and the best amount of contrast to get good image
- What is the best technique for getting a good enhanced pituitary gland, in addition to a clear image for any abnormality
Literature review
Imaging the hypothalamic pituitary axis is often based on specific endocrine testing that clinical signs and symptoms suggest (Marziallu et al. 2004). The excessive loss or deficiency of hormones characterizes endocrine disorders with excess hormones diagnoses under conditions that ordinarily suppress secreted hormones. The endocrine deficiencies are diagnosed based on hormone measurements or under stimulating conditions. The psychological role of a particular hormone defines the specific clinical syndromes of hormonal disorders. The hypothalamic pituitary axis included two separate neuroendocrine organs of the posterior and anterior systems (Marziallu et al. 2004). The posterior pituitary glands include anoxal terminations of neurons with cell bodies located in the hypothalamus. The cells secrete the principle oxytocin and vasopressin hormones, with the hypothalamus participating in complexly mediating anatomical functions. Structural cases include obesity, hypersomnia, insomnia, anorexia, and hyperthermia. The imaging of the patients often is unrewarding with the exception of children with relatively common diencephalic syndromes.
The pituitary adenoma is the most common lesion of the pituitary gland that secretes prolactina and gonadotrophins (Suzuki et al. 2005). In premenopausal females, prolactinomas is commonly present as microadenomas. The elevation of prolactin is non-specific and resulting from various medical, neurologic, and pharmacological causes as well as pituitary adenoma according to the level of serum hormones. In males, prolactinomas is entirely asymptomatic until visual symptoms occur because of the compressed chiasm. It may also result in hypogonadotropic hypogonadism where libido is lost and impotence is rampant (Suzuki et al. 2005). The larger lesions manifesting clinical acromelagy in the gland are the tumors that secret growth hormones. Given the actual onset of deformity, the tumors may be present for many years and grow substantially. Excessive growth hormone may result in gigantis, before puberty with thyroid stimulating hormone and adrenal corticotrophic hormone secreting tumors present in small sizes because of the apparent hormonal product that is more rapid.
Other lesions such as hamartoma may result from exhibit precocious puberty as well as other neurologicla symptoms (Suzuki et al. 2005). MR imaging is a good indicator in patients with endocrinologically conformed precocious puberty, particularly in instances, where neurological symptoms are present and pressing in their development. Other common lesions affecting the endocrine systems include Rathke’s cleft cyst, maningioma, cranio-pharyngioma, and germ line tumors. The scarcoid as well as other inflammatory processes occur in sella and supreasellar regions (Suzuki et al. 2005). Pituitary apoplexy is a result of pituitary hemorrhage exhibited by visual loss and headache opthalmoplegia. A pituitary macroadenoma with hemorrhage is shown in the figure below. During the postpartum period, pituitary infarction is often common, with hypophysitis being the uncommon disorder resulting from endocrine disturbances as well as other symptoms. The most useful test for the highlighted indications is the thin section multiplanar imaging before and after contrasting administration (Suzuki et al. 2005). Supplemental techniques include CT angiography, MR angiography, petrosal sinus sampling, and the direct conventional angiography.
Figure: Pituitary macroadenoma with hemorrhage. The arrow in Axial T1W (a) and T2W (b) images show intratumoral hemorrhage (Chaudhary & Bano, 2012)
Intravenously contrasting CT depicts the pathology with large unsettled sella to facilitate the diagnosis of neuroendocrine abnormality (Suzuki et al. 2005). The CT often detects pituitary microadenomas and well as macroadenomas. However, a difficulty has been noted in distinguishing tumor from optic chiasm or diagnosing the cavernous sinus invasion. Further, it is possible to confuse cystic suprasellar masses with the normal CSF. Other added disadvantages include artifacts arising from dental amalgam, the difficulties associated with obtaining reliance enhancement of contrast, and the out of depth positioning of direct limit unitility appearing in coronal scans. Experienced radiologists may find the technique useful, though interpretations are often difficult to make despite the technique being excellent.
According to Rotte et al. (2014), knowledge of the vascuar anatomy guides surgery with the sellar lesion displacing or encasing the carotid arteries as well as intracranial vessels. Interventional neuroradiology procedures are planned based on CT, MR, or conventional angiography. The petrosal sinus venous sampling is used in cases where a definite excess of the pituitary hormone is present, where medical management has failed, where sectional imaging is negative, or where an equivocal surgery is planned. Angiography is reserved for patients with known vascular pathology based on clinical as well as radiological findings (Rotte et al., 2014). Aneurism is an important vascular lesion of the paraselar region although they are rarely present in endocrine disorders. Where significant hormonal levels are present in the vessels under study, tumor localization often appears accurately. Rotte et al. (2014) note complications that may only arise in inexperienced hands, though uncommon.
A significant limitation of CT and MR imaging of the pituitary gland is endocrinal findings suggesting microadenoma as a false-positive examination. Given that endocrine studies confirm that lesions are present, and first line therapy is medical, a false-negative examination is less likely to have any affects upon conclusion of chiasmatic compressions. Nearly twenty percent of the population has incidental malfunctioning adenomas, making the probability of diseases high in the target population where a positive imaging is relied on for planning surgery. Additional problems arise because of the varying sizes of the pituitary gland, which respond to the changes in physiological hormones. The gland enlarges in puberty or pregnancy, with the pituitary hyperplasia simulating the pituitary adenoma in patients. The problems also arise in pituitary imaging, with over 29% of patients of normal subjects demonstrating a bright posterior pituitary. These limitations spark interest on the role of RMI in diagnosing the pituitary gland.
Methodology
The present paper employs the systematic review methodology to understand the documented trends in imaging modalities for the pituitary gland. The systematic review methodology is most proffered as a key element in research because it begins from a clearly formulated question, identifies related studies, appraises evidence quality, and summarizes the evidence using an explicit methodology (Khan, Kunz, Kleijnen & Antes, 2003).
The healthcare question
The present review seeks to cover the following areas on the role of MRI imaging in diagnosing the pituitary gland.
- The anatomy of pituitary gland
- The common pathology of pituitary gland
- The best sequences and facts for imaging the pituitary
- The role of injection contrast in diagnosing microadenoma, alongside the best amount of contrast to get good image
- The best technique for getting a goof enhanced pituitary gland, in addition to a clear image for any abnormality
Linking search criteria to the five questions
The population in the studies to be reviewed is not limited to any specific age, but is left open to general categories such as children, adults, pregnant women, women in late menopausal children, and children in puberty. The interventions and exposure applied in imaging the pituitary gland focuses mostly on magnetic resonance imaging, but leaves room for other imaging modalities that may help trace advancements in pituitary imaging. The outcomes of the study will include successful and unsuccessful imaging techniques of the pituitary gland as well as limitations in various models. The study design is not limited to a particular group, but leaves room for the integration of any peer reviewed article from 2000 to 2015.
Identifying relevant publications
To capture the most relevant studies, various medical and scientific databases were searched to identify the primary studies relating to MRI imaging of the pituitary gland. The electronic search was supplemented using a hand search of various internet search engines to provide related evidence. The relevance of the identified studies was examined, and the irrelevant studies excluded. The remaining bulk of papers were assessed to select primary studies covering the pituitary gland of human beings and MRI related imaging. Out of the near 167-idnetified studies relevant to the study question, only fifteen with MRI imagery were picked.
The studies were then assessed for quality based on the noted inclusion criteria. Summarizing the evidence from studies with various designs and quality was not easy with the differences between study results investigated to produce the best range of results. The paper was restricted to a narrative summary of findings with the association between magnetic resonance imaging and the pituitary gland being detailed in studies. Although other imaging modalities were also identified, the focus was on magnetic resonance imaging. The results had to be interpreted cautiously with the efforts in searching the large number of databases providing safeguards for the missing relevant studies. The evidence compiled in the study was evaluated as good.
Specific search strategy
A detailed specific strategy was used to analyze studies from MEDLINE, Google Scholar, ProQuest, Cochrane Central, MedPub, and CINAHL databases. The search strategy corresponded the criteria outlined below
- A search was conducted across databases, using the title to find abstracts and full studies. The index titles of the described articles were also identified to help in locating the appropriate studies.
- The search also included specific works and index terms used in the numerous databases to find the appropriate studies
- The reference lists were also analyzed to locate additional studies for user in the review.
- Key terms used in the study include Magnetic resonance imaging, neuroimaging, pituitary, recent advances, anterior and posterior lobe, hypothalamic hormones
MRI sequences of the pituitary gland
Date from the bulk of the studied show that Magnetic Resonance Imaging is a preferred imaging procedure for the pituitary as well as other parasellar regions, because of its qualities such as excellent spatial resolution, multiplanar capabilities, its ability to assess dynamic contrast enhancement and lack of ionizing radiation (Wisse et al. 2012; Chaudhary & Bano, 2012; Marziallu et al. 2004). MRI has grown into a clear procedure that most radiologists opt for in imaging the pituitary gland. Imaging using a high resolution MRI takes place in an environment where the high field strength magnet equals to the 1.5 or 3 Telsa unit (: Suzuki et al. 2005). Whereas the suprasellar content is depicted on low and medium strength field units, confidence in the diagnosis requires a high resolution obtainable in high field strengths only. For the high spatial detail, thin slices of preferably 2mm, a fine matrix of 256 x 256/512, and a field view of 16 to 18 cm are needed. The single intraglanular pathology measures 1-3mm with the gap resulting in a missed pathology where contiguous or no gap sections are required. Additionally, high signal to noise ratio is needed to deter obscurity for the small lesions. The widely employed pulse sequence is the conventional T1 weighted image in the coronal and sagittal planes with repetitive and echo time. Good signal to noise ratios result from two or four excitations, with more than five excitations increasing imaging time, and the possibility of patient movement (Tanase, Ogrezeanu & Badiu, 2011). Placing inferior saturation bands diminishes flow related artifacts on post-contrast sequences.
Whereas T2 imaging is often not necessary, coronal fast spin-echo T2 weighted images may improve tissue differentiation as shown below. Coronal and sagittal images are often sufficient, with axial imaging reserved for cases with large masses whose lateral extension need to be defined. Authors (Marziallu et al. 2004; Suzuki et al. 2005; Gutenberg et al., 2009), advocate for dynamic scanning of lesions for increased sensitivity to pituitary lesions. The authors propose that with fast scanning occurring during early infusion of contrast, it is possible to detect the differentiation between normal and neoplastic tissues due to the different dynamics of contrast enhancement. Investigations on the differences between dynamic and conventional sequences find superior sensitivity in detecting microadenomas. The largest of the series found that dynamic MRIs exhibit improved sensitivity, even as they exhibit decreased specificity when compared to conventional MRIs. However, it is important to note that the study was limited to the 1.0 Tesla magnets unlike contemporary studies that use no less 1.5 Tesla magnets.
The anterior pituitary gland generates a heterogeneous signal with a close isointense to the cortical brain on non-contrast T1 weighted images. The signal moving throughout the entire pituitary increases with contrast administration. In fetuses at the third trimester, infants between 2 to 3 months, and pregnant women, and, the anterior pituitary come across as a hyperintense. This is secondary to hyperplasia of prolactin cells, which respond to the placental estrogen. It is easy to recognize the cavernous sinuses because of their relative hypo-intensity to the pituitary gland and contiguous brain that makes them easy to recognize. These also show significant enhancement with contrast, even though the medial dural margin of the cavernous sinus is not visible as a separate structure. Conversely, the lateral dural wall is well defined with the intracavernous neutral structures exhibiting lower intensity than the sinus. The rapidly flowing carotid arteries result in a signal void clearly defining the vessels in non-contrast scans. Since the cavernous sinuses enhance the pituitary gland, equally the margin between the two is less conspicuous on contrast-enhanced scans despite the carotid arteries as well as the neural structures remaining visible to the rapid flow and low signal of the normally enhanced structures.
Results of the study
The bulk of literatures studied reveal that pituitary imaging is important in confirming diagnosis of pituitary lesions and in differentially diagnosing the sellar lesions. For instance, Chaudhary and Bano (2012) assert that the use of plain skull radiographs in diagnosing sellar and parasellar pathologies is limited today because they are poor at delineating soft tissues. The radiographic size of the sellar does not sensitively indicate the abnormality in the pituitary gland as empty sella enlarges size. Therefore, plain radiographs are replaced by cross-sectional imaging techniques such as magnetic resonance imaging or CR scans. Another author, Drouin (2010) adds that CT scanning is useful in examining soft tissue calcification, especially in contraindications of the MRI although it is not used in most sellar and parsellar lesions. These include patients with pacemakers and those with metallic implants in the brain or the eye. However, the authors note the drawbacks of using CT scans link to less optimal soft tissue contrast and exposure to radiation, which limit its use in CT scanning for evaluating pituitary lesions (Chaudhary Bano, 2012; Drouin, 2010).
MR imaging works towards obtaining high spatial resolution image with a sensible signal to noise ratio. It is necessary to locate the gland separately from lesions where possible. Initially, precontrast T1 and T2 weighted spin echo coronal and sagittal sections were applied in gaining high-resolution images. The small FOV of 20 X 25 cm, thin slices of 3mm and a high-resolution matrix of 256 X 512. The dynamic and precontrast images combine with delayed scanning of thirty to sixty minutes to achieve a single study of optimum imaging. Whereas non-enhanced MR imaging detect most adenomas, mircoadenomas are only visible after contrast is injected (Chaudhary & Bano, 2011). Dynamic contrast MRI has evolved as the best imaging tool in evaluating pituitary adenomas. Dynamic studies require three dimensional Fourier transformation gradient echo as well as fast turbo spin echo sequence. The process requires a dose of 0.05 mmol/kg of gadolinium injected through the veins. After injecting gadolinium through the veins, the coronar plane produces images every ten seconds (Chaudhary & Bano, 2011). This enhancement occurs in the pituitary stalk, to the pituitary tuft and to the centrifugal pacification of the anterior globe. The imaging modality obtains maximum image contrast between the microadenoma and the normal pituitary tissues thirty to sixty seconds after the bolus is injected via the vein.
According to Chaudhary and Bano (2011), most microadenomas appear as non-enhancing lesions that enhance the pituitary gland intensely. The peak of an enhanced image is the 60-200 mark, which follows a marked enhancement of the normal pituitary gland and persists for longer durations. A delayed scan of 30 to 60 minutes is retrieved following contrast injection reversing of the image contrast obtained at 30 to 60 seconds using dynamic scans. This is a result of the fading contrast from the normal pituitary gland, which later diffuses and into the microadenoma. The fading contrast stands out as a hyper-intense focus. The enhancement of the adenoma is documented widely in studies, even as the micro-adenoma is enhanced before the anterior lobes (Kakite et al 2011; Portocarrero-Ortiz et al. 2010). This occurs out of the pituitary adenoma supplying arterial blood directly just as the posterior pituitary lobe. Adding the dynamic sagittal plane images of the routine to coronal studies increase the possibility of detecting microadenomas. Dynamic contrast MRI helps in evaluating pituitary microadenomas and assessing macroadenomas. The modality also helps in assessing the invasion of the cavernous sinus using the macroadenomas. Last, it helps differentiate the recurrent tumors occurring in postoperative tissues. Macro-adenomous exhibit maximum contrast when the lesion to gland is scanned using unenhanced imaging. Still, the image contrast begins diminishing as soon as the contrast enhancing agents reaches the pituitary gland.
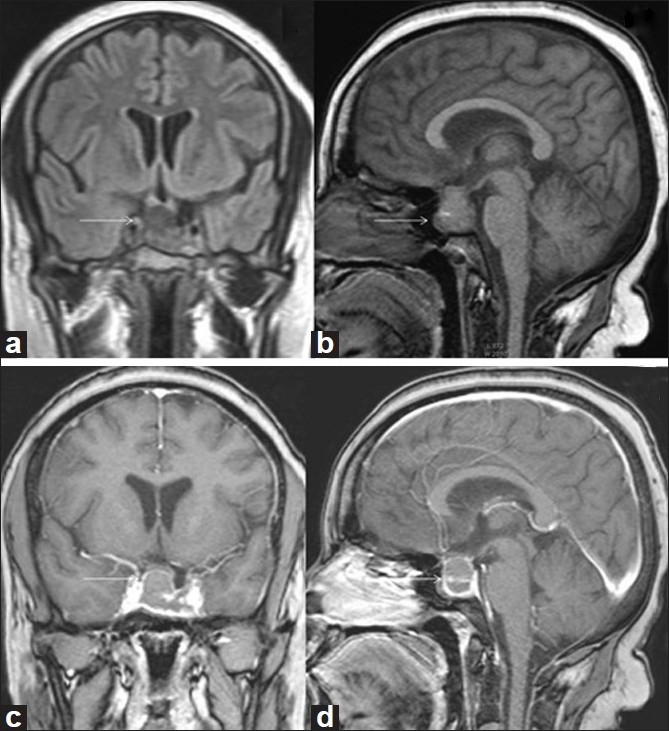
Tesla MRI is hailed for its stronger magnetic strength, which allows for improved image quality (Pinker et al. 2005). It also allows for spatial resolution under the conditions where the normal and abnormal tissues are subtly different. A recent study on the operative localization of the pituitary micro-adenomas reveals that, Cushing’s disease is better identified using the 3T and not 1.5T MRI. A cross comparison of 3T and 1.5T MRI imaging is shown in the figure below. The results are expended despite some lesion missing on 3T MRI. Further studies using the 3T MRI on the invasion of the cavernous sinus by the adenomas across 42 patients reveals that T1 weighted magnetization prepares the rapid acquisition gradient echo sequences to delineate the cavernous sinus invasion while using the intrastellar as compared to standards MRI techniques (Pinker et al. 2005). The only limitation was that the study did not compare the 3T with 1.5T MRIs with respect to the type of tumor. The use of 3T MRI is evolving revolutionarily, meaning that further refinement using new sequences to confirm the presence of microadenomas that fail to produce contour abnormalities is required (Rotte et al. 2014). The evolution is also important in identifying the number and location within the glands. The pieces of information are important for planning surgical management of microadenomas. Further, having knowledge of the sequences in normal pituitary gland volumes makes the normal appearance of imaging sequence important in diagnosing gland and infundibulum lesions (Pinker et al. 2005). Accurately assessing the stalk as well as the occurring subtle changes in the volume of the gland is better evident using 3T than 1.5T MRI.
Figure: Contrast on the 1.5T MRI and (B) 3T MRI (Chaudhary & Bano, 2012)
Whereas it is possible to delineate plain and contrast enhanced MR sequences, classical MRI cannot differentiate between secreting and non-secreting adenomas. Moreover, the role of MRI in evaluating residual tumors in most post-operated case remains limited (Pinker et al. 2005). A recent advancement in the field of pituitary gland imaging is Magnetization transfer (MT). Magnetization transfer is applied in pre-and-post-operative assessment of pituitary adenomas, especially in patient’s diagnoses with hyperprolactinemia. The tissue contrast of magnetization transfer imaging depends on concentrated macromolecules. The contrast is further quantified using the magnetization transfer ratio. For patients with hyperprolactinemia, the ratio of adenomas that secrete prolactin is higher, unlike the value of the normal pituitary gland (Pinker et al. 200). Conversely, non-secreting adenomas exhibit lower magnetic transfer ratios compared to normal pituitary glands, thereby demonstrating the low signal of MT images. The difference between the two adenomas is significant because of their separate management. Further, most prolactinomas are treated using medications while non-secreting adenomas are managed using surgery (Pinker et al., 200).
Other reported imaging modalities
The magnetization transfer technique is also used in post-operative assessment and for following up patients with pituitary adenomas. This happens in particular cases, where the classical MRI is negative for residual tumors. Increasing the values of magnetization transfer ratios is suggested for persisting adenomas tissue (Kim et al., 2004). Additional prospects of magnetization transfer imaging includes pituitary disorders, for instance, pituitary insufficiency, and precocious puberty. Diffusion-weighted imaging used in detecting acute pituitary infarction has been analyzed to reveal pituitary apoplexy, which often results in hemorrhages of pituitary gland infarctions linked to high mortality and morbidity rates. It is further documented that diffusion-weighted imaging (DWI) helps in the early diagnosis of acute pituitary infarction where intervention is expended in a timely fashion to produce excellent outcomes. Imaging may diagnose pituitary infarction where peripheral contrast enhancement of intrasellar masses is present on contrast-enhanced images (Kim et al., 2004). It is also diagnosed when the contrast enhancement is made on DW image lesions. Acute pituitary infarctions on diffusion-weighted imaging mimic pituitary hemorrhage or abscess. Studies show that diffusion-weighted imaging and apparent diffusion coefficient are able to characterize tumor components present in the adenomas and to offer consistent information about micro-adenomas. According to Kim et al. (2004), most of the microadenomas are easily resectable and soft because they use minimal invasive endoscopic techniques and require intensive surgery. Therefore, the knowledge of consistent adenomas helps clinicians in planning proper surgical techniques for resection. When taken on MRI, the sift microadenomas are inhomogenous, hyperintense or isointence on T2 weighted images. When appearing on T1 weighted images, they are hypointense with an enhanced contrast on post gadolinium images. The lesions demonstrate high signal intensity on DWI imagining and low ADC values. Microadenomas in intermediary groups are inhomogenous and hyperintense on T2 weighted images. They also appear as iso-or-hypointense in T1 weighted images, meaning that they are markedly enhanced after administering intravenous contrast agents. On DWI, the lesions demonstrate high signal intensity with the ADC value appearing higher than soft adenomas and lower than hard adenomas. Studies show that hard adenomas are inhomogenous, hyperintense on T2 weighted images, and hyperintense of T1 weighted images to exhibit enhanced intravenous contrasting administration. The tumors demonstrate low signal intensity on DW images and with relatively high ADC values. DWI should be included in routine assessment of macroadenomas to allow for easy planning of surgery.
Proton MR Spectroscopy assists in differentiating the types of lesions that involve the pituitary-hypothalamic axis (Kim et al., 2004). The proton MR spectropy of hypothalamic gliomas have a marked increased presence of choline and a decrease in N-acetyle aspartate., However, craniopharyngiomas and germiomas often exhibit dominant lipid peaks. It is important to note that pituitary adenomas show the choline peak only with no metabolites present in intratumoral hemorrhage. This is because hemosiderin worsens the homogeneity of the magnetic field. Findings of MR spectroscopy include decreased N-acetyle aspartate. Additional findings include increased myo-inositol as compared to grain matter.
Whereas MR imaging remains the most appropriate choice for evaluating pituitary tumors, numerous situations where positron emission tomography (PET) scans provide valuable information exist in assessing pituitary tumors (Kim et al., 2004). The Fluorine-18 fluorodeoxyglucose-positron emission tomography (FDG-PET) is also applied as a differentiation tool between residual and recurrent tumors from post operative changes as well as radiation injuries. The increased uptake of Fluorine-18 fluorodeoxyglucose exists in residual tumors while postoperative change so radiation injuries are evidence of decreased FDG uptake. The FDG-PET also helps in differentiating macroadenomas from sellar or suprasellar masses. Imaging modalities identify macroadenomas as foci of increased FDG uptake near the sella region with the uptake high in non-functional adenomas. The FDG-PET also stages and monitors the diseases to allow for better planning of treatment options.
Another recent advancement on pituitary imaging, the single-photon emission computed tomography (SPECT), is not promising. SPECT uses iodine 123-labeled dopamine D2 receptor antagonist iodobenzamide (IBZM) to evaluate pituitary tumors. A study on 15 patients having pituitary tumors show that IBZM-SPECT is poor at visualizing macroadenomas secreting growth hormones as well as prolactin. Most recently, pituitary imaging has advanced towards the use of interoperative MRI (IMRI) because it provides better visualization of the intra-and-parasellar anatony (Suzuki et al. 2005). This facilitates the complete resection of the tumor. Using a plasma screen alongside the IMRI is promising in the near future for its value in creating high quality images. These demonstrate never compression and residual tumors. Similarly, the intraoperative real-time ultrasonography used in surgical management of the endoscopic pituitary help in localizing intracranial neoplasms, especially in the deep skull base lesions. Ultrasonography also assists in guiding biopsy needles as well as aspirating pituitary lesions (Suzuki et al. 2005).
Marziallu et al. (2004) consider the 3D-MRI a reliable tool in evaluating anatomical volumes of the pituitary gland. The tool is an enhancement of the 2D-MRI used in volumetric measurements of the anterior pituitary gland in the past, which has inherent inaccuracies. Where the 3D-MRI was implemented in obtaining the accurate volumes of anterior gland using coronal sections of the 3D-MRI, studies show that a progressive growth of the gland was evident, meaning that it aided in evaluating patients with neuroendocrine diseases, especially growth hormone deficiency (Marziallu et al. 2004). Although the MR imaging modality reveals morphological abnormalities such as posterior pituitary exposure, stalk agenesis, and pituitary hypoplasia, detecting the causes of growth hormone deficiency (GHD) resulting from gland hypoplasia has proven difficult. When the affected gland sizes are compared with the database of psychological volumes, diagnosis can be improved. Previous studies measuring the sizes of the adenohypophysis through indirect methods such as calculating the biodimensional values of MRI have been hampered by the reduced reliability of these methods because of the morphological variations of the pituitary gland. The recent 3D-MRI techniques offer a reliable method for identifying anatomical volumes across adults and children.
Conclusion
From the investigation, it is plausible to conclude that the MRI is the best mode of evaluating hypothalamic pituitary-related endocrine diseases. MRI does not help in differentiating the diagnosis of the lesions, but offers useful information on the relationship between the gland and the nearby anatomical structures to help in planning medical or surgical strategies. DWI’s should be applied routinely in assessing macroadenomas for plan for the best approach to conducting surgery. Magnetic resonance imaging produces highly detailed images of midline brains structures, making it a chosen technique in neuroradiological evaluation of patients with pituitary gland problems. MRI reveals the structure and abnormalities associated with the pituitary region. For instance, the MRI reveals incidences of structural and morphological abnormalities in the pituitary region. The pituitary gland is an essential organ that plays a critical role in the growth, metabolism, and reproductive function of humans. Many diseases affecting the pituitary gland result in clinical, endocrinological, and neurological consequences. The consequences are classified as infectious, inflammatory, posttraumatic, physiological, congenital, and neoplastic.
MR imaging offers an excellent non-invasive evaluation of the pituitary gland as well as the hypothalamus. It is the only reliable technique capable of producing a useful image of the hypothalamus. MR imaging depicts the anatomy of the pituitary gland accurately and non-invasively. Where the gadolinium is added, diagnosis of the mircoadenoma is facilitated, and confidence for diagnosing the cavernous sinus invasion increased. Whereas the body landmarks are often difficult to demonstrate, the sphenoid sinus mucosal signal intensity allows radiologists to assess the septa to achieve operative planning. Visualizing the vascular parasellar structures is important for cases such as aneurism.
References
Chaudhary, V. & Bano, S. (2011). Imaging of the pituitary: Recent advances. Indian J Endocrinol Metab, 15(3), 216–223.
Chaudhary, V. & Bano, S. (2012). Imaging of pediatric pituitary endocrinopathies. Indian J Endocrinol Metab, 16(5), 682–691.
Drouin, J. (2010). “Pituitary Development.” In S. Melmed. “The Pituitary: Third Edition.” Waltham, USA: Academic Press.
Gregory, L. C. et al. (2013). Structural pituitary abnormalities associated with CHARGE syndrome. The Journal of Clinical Endocrinology & Metabolism, 98(4), E737-E743.
Gutenberg A. et al. (2009). A radiologic score to distinguish autoimmune hypophysitis from nonsecreting pituitary adenoma preoperatively. AJNR Am J Neuroradiol., 30, 1766–72
Kakite S. et al (2011). Three-dimensional gradient echo versus spin echo sequence in contrast-enhanced imaging of the pituitary gland at 3 T. Eur J Radiol 79, 108–11 2.
Khan, K. S., Kunz, R., Kleijnen, J. & Antes, G. (2003). Five steps to conducting a systematic review. J R Soc Med., 96(3), 118–121.
Kim, J. E.et al. (2004). Surgicaltreatment of symptomatic Rathke cleft cysts: clinical features and results with special attention to recurrence. J Neurosurg, 100, 33-40.
Marziali, S. et al. (2004). Evaluation of anterior pituitary gland volume in childhood using three-dimensional MRI. Pediatr Radiol, 34, 547–551.
Maya, M. M. & Pressman, B. D. (2010). “Pituitary Imaging.” In S. Melmed. “The Pituitary: Third Edition.” Waltham, USA: Academic Press.
Pinker K, Ba-Ssalamah A, Wolfsberger S, Mlynarik V, Knosp E, Trattnig S (2005) The value of high-field MRI (3 T) in the assessment of sellar lesions. Eur J Radiol 54, 327–334.
Portocarrero-Ortiz, L. et al. (2010). A modified protocol using half-dose gadolinium in dynamic 3-Tesla magnetic resonance imaging for detection of ACTH-secreting
pituitary tumors. Pituitary, 13, 230–235.
Rotte, A. A. J. et al. (2014). Feasibility of high-resolution pituitary MRI at 7.0 tesla. Eur Radiol, 24, 2005–2011.
Suzuki, M. et al. (2005). Dynamic MR imaging for diagnosis of lesions adjacent to pituitary gland. European Journal of Radiology, 53, 159–167.
Tanase, C., Ogrezeanu, I. & Badiu, C. (2011). Molecular Pathology of Pituitary Adenomas. Netherlands, Amsterdam: Elsevier.
Wisse L. E. et al. (2012) Subfields of the hippocampal formation at 7 T MRI: in vivo volumetric assessment. Neuroimage, 61, 1043–1049
Here are some the nursing writing services we provide:
– Nursing Dissertation Writing Services
– Nursing Paper Writing Services
– Nursing Essay Writing Service
– Nursing Assignment Help
Our excellent biology writing help team of academic writers provides a wide range of biology academic help not limited to:
– Biology Assignment Writing Services
– Biology Assignment Help
– Biology Dissertation Writing Service
– Biology Essay Writing Service