Microbiology Staining
Making Negative Stain
In negative staining, acidic stains such as nigrosin or India ink are used. These stains contain the negatively charged chromogen that doesn’t penetrate the cells due to the negative charges that are found on the cells (Aryal). The following is a procedure for negative staining;
- A small drop of nigrosin is placed at the periphery of a clean slide.
- A substantial inoculum of the cultured bacteria put into the nigrosine via aseptic method and mixed.
- The slide is slipped against the drop of the suspended bacteria at 45o so that the drop spreads out.
- The slide is pushed away from the bacteria and a thin smear form. The slide is air-dried; no heat-fixation.
- A microscope is used to view the slide while under an oil immersion (Aryal).
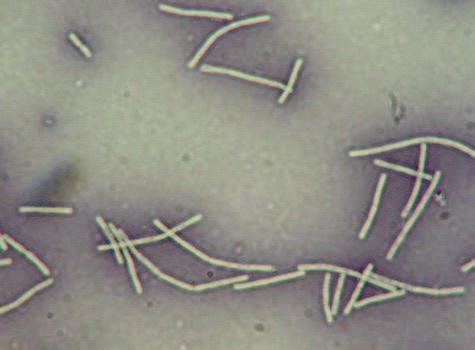
Figure 1. Negative staining of Bacilli: Magnification x1000
The negative stain outcomes have the cells in their natural shapes and sizes because there was no distortion of the cells by either chemical or heat. Bacterial cells that are not easily stained can be seen, the instance spirilla bacteria. The slides need to be handled carefully since the bacteria live; they were not destroyed through heat fixation (Aryal).
Making Gram Stain
A gram stain is the most common method used to identify bacteria. There are mainly two types of bacteria; gram-positive and gram-negative. The gram staining technique uses a crystal violet stain that produces a purple-blue color on the entire smear. Iodine in the gram serves as a mordant. It makes the crystal violet stain enter and bond to the gram-positive bacterial cell. Acetone-alcohol mixtures used as decolorizer washes the stain off from all smear parts except the gram-positive cells. Safranine is the counter-stain that allows staining of everything in the smear colonized by the gram-negative cells (Microscope.com). The basic steps for gram staining are as follows;
- The bacterial smear is applied on the slide, air-dried, and heat-fixed by passing it over a flame.
- Five drops of Hucker’s crystal violet are added to the culture and given 1 minute to settle. After the bacteria is stained, water is used to wash briefly, and the excess is dropped off.
- Five aqueous iodine are added to the culture and given thirty seconds for settling. The slide is washed briefly with water, and the excess is shed off.
- The slide is tilted, then decolorized using aqueous acetone-alcohol up to when the purple color stops to come out. Immediate washing with water (within 5 seconds) then the excess is shed (shaken) off.
- Five drops of safranine O is added and given 1 minute to settle. Brief washing using water was done.s
- The slide is observed under oil immersion at x400 and x1000 magnification.
If the organism is gram-negative, an entirely red slide will be observed. If the bacteria are gram-positive, an entirely purple slide will be observed. It should also be noted that the outcome is influenced by the integrity of the procedure and the reagents. Therefore, one may consider changing the reagents or staining method to get desirable outcomes. For instance, a gram stain for a swab sample collected from the mouth should indicate both gram-positive and gram-negative bacteria. If such a stain indicates only one type of bacteria, it means that either the straining procedure or the reagents (or both) were not efficient (Microscope.com).
Our excellent biology writing help team of academic writers provides a wide range of biology academic help not limited to:
– Biology Assignment Writing Services
– Biology Assignment Help
– Biology Dissertation Writing Service
– Biology Essay Writing Service