A REVIEW ON CD MARKER PANEL FOR CHARACTERIZATION OF RAT AND MOUSE ADIPOSE STEM CELLS VIA FLOW CYTOMETERY
Abstract
ADSCs are involved in healing and repairing of various body tissues. Presence of surface antigens on ADSCs marks their ability to bind with specific CD markers. Flow cytometry is a common method used in laboratories to validate and identify different cell types, including ADSCs. In this study we reviewed published articles in which rat and mouse ADSCs were characterized via flow cytometry. Comparing the phenotypic characterization results of these studies, we seek to provide a comprehensive map of which CD markers are most common and effective for the characterization of rat and mouse ADSCs. Different CD markers are used for characterization of ADSCs in rat and mouse amongst which CD90 and CD44 are the most common. This review may contribute to demonstrate minimal standard via flow cytometry for the characterization ADSCs of rat and mouse This review aims to provide the detailed overview for the use of most common CD markers for characterization of adipose stem cells both in-vivo and in-vitro to get desired and predictable therapeutic effects.
Graphical Abstract
Keywords: ADSCs; Adipose tissue; Flow cytometry; Stem cells, CD marker.
Introduction
After the discovery of mesenchymal stem cell (MSC), extensive researches have been conducted to highlight the unique properties of these cells. Special attention was given to study and examine the characteristic morphology of MSC along with their self-renewal property, and capability to differentiate. Stem cells have gained adequate importance in the clinical and medical field. For the study of stem cells, Adipose derived stem cells abbreviated as ADSCs prove to be exceptional source of raw material. Adipose derived stem cells (ADSCs) originate from adipocytes. They are able to differentiate into many cell types and are potentially unique by possessing multiple characteristics. Many basic and clinical researches were organized highlighting the role of ADSCs in regenerative technologies, biomaterial development and cosmetic applications [1]. ADSCs serve as the best choice to conduct research because of their unlimited availability, uncomplicated isolation from body and self-renewable characteristic [2].
Stem cells isolated from rat or mouse are generally used for researches in the field of tissue engineering. Whereas, researches reveal noticeable differences in mouse/rat mesenchymal stem cells and that of human beings. There is no defined marker panel for characterizing these cells. To evaluate cell composition in a sample and expression of surface markers, flow cytometry is the most widely used method [3].
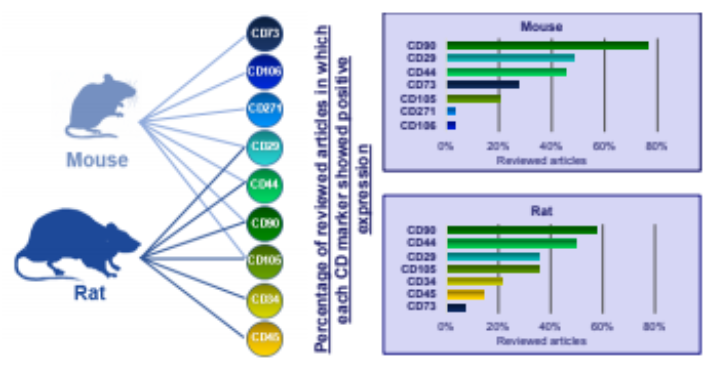
Researchers have used rodents as practical model to access host tolerance and to evaluate the performance in biological transplants. Rats were used in almost 82% of the researches till now due to their small size, availability and simple experimental administration [3]. Multipotent mesenchymal cells have ability to adhere to tissue culture polystyrene (TCP). ADSCs possess surface antigens that bind to wide panel of CD markers. These include a variety of positive markers which include CD90, CD23, CD13, CD29, CD34, CD44, CD73, CD166, CD10, CD49e and CD59 whereas negative CD markers include CD14, CD11b, CD31, CD45, CD34, CD19, CD56 and CD146 [4]. Comparing the phenotypic characterization results of these studies, we seek to provide a comprehensive map of CD markers that are most common and effective for the characterization of rat and mouse ADSCs via flow cytometry. The most commonly used CD markers for characterization of adipose stem cells in rats are CD90, CD29, CD44, CD105, CD73, CD271, CD106 and mouse are CD90, CD29, CD44 and CD73, CD45, CD105 along with CD34, CD156, CD41 and CD46
This review has summarized the available data that leads to characterization of ADSCs. The most common panel of CD markers for characterization of rat and mouse adipose stem cells via flow cytometry will allow the most reliable method to identify ADSCs both in vitro and in vivo. The main aim of this review is to highlight CD markers for detection of ADSCs via flow cytometry from mouse and rat that differentiates into different cell lineages both in-vivo and in-vitro. This review may enhance the notion that ADSCs are involved in healing and repairing of various body tissues.
Discussion
Stem cells are the raw cells of living organisms which have the ability to regenerate. One of the main sources of stem cells is adipose tissue. In recent studies, adipose tissue contains pluripotent and pluripotent stem cells that can regenerate and differentiate into a variety of specialized cell types. Adipose stem cells (ASC) are mesenchyme stem cells (MSC), derived from a large amount of adipose tissue, adhered to plastic culture flasks, can be expanded in vitro, and have the ability to differentiate into a variety of cell lineages. [5]. There are two main sources of stem cells; adult body tissues and embryos. Scientists are additionally working on ways to develop stem cells from other cells, utilizing genetic “reprogramming” techniques.
Scientists discovered a method to extract embryonic stem cells from early mouse embryos in 1981 nearly 30 years ago. The detailed study of mouse stem cell biology led to the discovery in 1998 of a method of extracting stem cells from human embryos and growing them in the laboratory [6].
The concept of stem cells was first proposed by researchers in the 19th century. They see such cells as the starting point of biological processes. Since the beginning of the twentieth century, stem cells have been regarded as the source of various blood cells. However, blood stem cells cannot be seen during this period, so many people suspect their existence. During the Second World War, the development of knowledge was promoted by identifying a restoration factor in the bone marrow, which was believed to help regenerate the blood system. Hematologists William Bloom and Leon Jacobson discovered this while studying the effects of radiation on the blood system, as part of their “Manhattan Project” and made it to make the first atomic bomb. In 1978, stem cells were also found in human umbilical cord blood. Three years later, scientists cultured the first embryonic stem cells from mouse blastocysts [7].
Characterization of pluripotent stem cells is required for the registration of stem cell lines and sanctions for an impartial and objective comparison of the results obtained when engendering multiple lines. It is consequently crucial to establish concrete, expeditious and reliable protocols to detect the hallmarks of pluripotency [8]. The first method used to characterize stem cells is by in vitro differentiation. One of the main sources of stem cells is adipose tissue. In recent studies, adipose tissue contains pluripotent and pluripotent stem cells that can regenerate and differentiate into a variety of specialized cell types.
Other technologies used for stem cell analysis and characterizations include:
- Flow cytometry:
Flow cytometry may be a widely used method for analyzing molecule present on cell membranes or inside the cell, expression, characterizing and defining different cell types in heterogeneous cell populations, and evaluating the purity of the separated subpopulations, and analysis of cell size and volume [9].
- Array-based analysis of transcriptomes:
Transcriptome Analysis is the study of the transcriptome, of the consummate set of RNA transcripts. [10].
- Immunocytochemistry:
Immunocytochemistry is a technique that’s utilized to anatomically visualize the localization of a categorical protein or antigen in cells. [11].
- Western blotting:
In this technique an amalgamation of proteins is disunited predicated on molecular weight, and thus by type, through gel electrophoresis. [12].
- Biomarker analysis (different types of stem cells are characterized by different combinations of markers): Biomarkers are distinctive biological characteristics which can be discovered and quantified in components of the body like the blood, serum or tissue. [13, 14].
Sundry studies are conducted to spot hematopoietic stem cells (HSCs) utilizing flow cytometry. The technique is primarily predicated on fluorochrome-conjugated antibodies against cell surface markers of HSCs and therefore the physiological properties of HSCs like high-efflux activity of certain fluorescent dyes [15].
Embryonic stem cells are distinguished by two distinctive properties: their pluripotency, and their competency to duplicate indefinitely. ES cells are pluripotent, that is, they’re ready to differentiate into all derivatives of the three primary germ layers: ectoderm, endoderm, and mesoderm [16].
In 1978, flow cytometry was first wont to identify stem cells. Len Herzenberg, an immunologist at Stanford University, pioneered the tactic of sorting cells using the principles of flow cytometry. He coined the term FACS-fluorescence activated cell sorter-to identify and count cells. The old term use for flow cytometry was pulse cytophotometry [17].
Flow cytometry is a popular cell biology technique that uses laser technology to count, sort, and profile cells in a heterogeneous fluid coalescence. Utilizing a flow cytometer machine, cells or other particles suspended during a liquid stream are skilled a laser beam in file fashion, and interaction with the light is quantified by electronic detection device as light scattering and fluorescence intensity. If a fluorescent label, or fluorochrome, is categorically and stoichiometrically bound to a cellular component, the fluorescence intensity will ideally represent the quantity of that particular cell component [18].
In 1968, Wolfgang Göhde of the University of Münster in Germany developed the first fluorescence-based flow cytometry technology (ICP 11), and it was first commercialized by a German developer in 1968/69. [19].
The research applications of flow cytometry include:
Immunophenotyping:
This technique identifies and quantifies populations of cells in a heterogeneous sample conventionally blood, bone marrow or lymph.
Cell Sorting:
The cell sorter is a specialized flow cytometer with the competency to physically isolate cells of interest into separate amassment tubes. The cytometer interrogates and characterizes each cell because it passes through the laser.
Cell Cycle Analysis:
Flow cytometry can analyze replication states utilizing fluorescent dyes to quantify the four distinct phases of the cell cycle. Along with determining cell cycle replication states, the assay can quantify cell aneuploidy associated with chromosomal abnormalities.
Apoptosis:
Apoptosis, or programmed cell death, is a mundane part of the life cycle of eukaryotic cells. Cells die for a spread of reasons: through necrosis, brought on by external physical and chemical changes to the cell or through apoptosis, a process during which cells initiate a “suicide” program through internally controlled factors. These two distinct types of cell death, apoptosis and necrosis, can be distinguished by flow cytometry on the substratum of differences in morphological, biochemical and molecular changes occurring in the dying cells.
Cell Proliferation Assays:
Cell proliferation assays are widely utilized in cell biology to quantify cellular metabolic activity in replication to stimuli such as magnification factors, cytokines and other media components. The flow cytometer can quantify proliferation by labeling reposing cells with a cell membrane fluorescent dye.
Intracellular Calcium Flux:
Cells interact with each other and their surrounding environment through signal transduction pathways. When these pathways are activated, membrane-bound calcium ion channels pump calcium into the cell and expeditiously increase the intracellular calcium concentration. The cytometer can monitor the flux of calcium into the cell and quantify the extent to which cells respond to the stimuli [20].
Flow cytometry is a high-speed technology by which single cells, cell aggregates, or minute multicellular organisms in liquid suspension can be relegated and viably sorted at authentic-time rates well in excess of thousands per second. Cell relegation by flow cytometry departs from automated microscopy in that cells are identified by a minimal set of scalar parameters without endeavoring to spatially resolve and analyze images [21].
Some of its major advantages are:
1) It sanctions the simultaneous analysis of different cellular features with high performance and reliability.
2) It enables the disunion of living cells on the substructure of marker expression or functional properties by fluorescence‐activated cell sorting.
3) A major advantage of this technique is its faculty to isolate rare cells, which is a prerequisite of identifying diminutive cell populations within the tumor bulk.
4) It is withal possible and can be achieved either by adding count check beads to the sample or by volume‐based flow cytometry.
5) Flow cytometry can be subsidiary in sub classifying AMLs without recurrent cytogenetic abnormalities [22].
In 2001, when Zuk isolated and identified adipose stem cells, he used adult stem cells from liposuction adipose tissue. Since then, flow cytometry has been used to characterize ADSCs in many studies. Although the CD nomenclature has only been established for human antigens [23]. it is also applied to antigens from other species. Furthermore, adipose tissue may have different biochemical profiles among species and may lead to various phenotypic results. Therefore, an extensive investigation of which CD Markers are suitable for each species, as well as a comparative CD Marker panel can both aide in the characterization of animal ADSCs via flow cytometry.
For the best export of the results, we decided to categorize our collected data into articles that refer to mice and those that refer to rats. In both rats and mice, we considered each subspecies separately. We did not find sufficient articles in the literature regarding the use of cytometry to identify stem cells in species other than those mentioned above and therefore we did not include them in this review.
As we are familiar that there are 82% of the researchers who have identified ADSCs in rats using flow cytometry due to their small size, availability and simple experimental administration. Table 1 shows the corresponding CD markers used while Table 2 shows common CD markers used in each subspecies separately. It was observed that no matter which subspecies is under study, common CD markers found for identification od ADSCs are CD90 and CD44 which give best results.
Table 1.1 CD positive and negative markers via Flow cytometry in different subspecies of rats
| Sr # | Specie | CD+ | CD- | Ref |
|
|
SD Rats | CD90, CD44 | CD34, CD45 | [24] |
|
|
CD29, CD90 | CD34, CD45 | [25] | |
|
|
CD44, CD105 | CD31, CD45 | [26] | |
|
|
CD90, CD44 | CD34, CD45 | [27] | |
|
|
CD29 | CD45, CD90 | [28] | |
|
|
CD105, CD90 | CD34, CD45 | [29] | |
|
|
CD29, CD44 | CD31, CD106, CD184, CD34, CD45 | [30] | |
|
|
CD29, CD90 | CD45 | [31] | |
|
|
CD90, CD29 | CD106, CD11b, CD45, CD49d | [32] | |
|
|
CD29, CD44, CD73, CD90 | CD 11b/c, CD45 | [33] | |
|
|
CD29, CD90 | CD45, CD34, CD31 | [34] | |
|
|
Sca-1, CD44 | CD45, CD11b | [35] | |
|
|
CD29, CD73, CD90 | CD34, CD45 | [36] | |
|
|
CD90, CD73, CD29 | CD34, CD45 | [37] | |
|
|
CD271, CD73 | CD45 | [38] | |
|
|
Wistar Rats | CD44, CD90 | CD34 | [39] |
|
|
CD73, CD90, CD105 | CD31, CD34, CD45, CD271 | [40] | |
|
|
CD44, CD90 | CD45 | [41] | |
|
|
CD90, CD29 | CD34, CD45 | [42] | |
|
|
Wistar–Imamichi | CD105, CD29, CD44 CD73, CD90 | CD34, CD45 | [43] |
|
|
VEGF, EGF, rPH Ki-67 | CD45 | [44] | |
|
|
Albino Rats | CD44, CD73, CD90 | CD45 | [45] |
|
|
Rattus norvegicus (Fischer 344 Rats) | CD90 | CD45 | [46] |
|
|
Lewis Rats | CD29, CD44, CD90 | CD11, CD31, CD45 | [47] |
|
|
CD90, CD105 | CD34, CD45 | [48] | |
|
|
CD90, CD73, CD105 | CD34, CD45 | [49] | |
|
|
CD29, CD44, Stro-1 | CD34 | [50] | |
|
|
CD44, CD90, CD106 | CD45, CD80/B7-1 | [51] |
Table 1.2 CD positive and negative markers via Flow cytometry in different subspecies of mice
| Sr # | Specie | CD+ | CD- | Ref |
|
|
Transgenic C57BL/6 mice | CD105, CD44 | CD45, CD34 | [52] |
|
|
CD29, CD90, CD105 | CD45 | [53] | |
|
|
CD146, CD34 | CD29 | [54] | |
|
|
CD90, CD105, Nanog SSEA-1, CD106, VEGFR-1 | CD34, CD45 | [55] | |
|
|
CD44, CD90.2 | CD34, CD45 | [56] | |
|
|
CD44, CD90.2 | CD34, CD45 | [57] | |
|
|
CD45, CD44 | Sca-1, CD90, CD105 | [58] | |
|
|
CD90, CD73 | CD31, CD45 | [59] | |
|
|
Sca-1, CD90, CD105 | CD45, CD34, HLA-DR | [60] | |
|
|
C57BL/10J and BALB/cJ | Sca-1, CD34 | CD45, CD31, CD144, CD146, flk-1 a | [61] |
|
|
CD44, CD90, CD105 | CD11b, CD14 CD34, CD45 | [62-64] | |
|
|
(GFP) transgenic mice | CD44, CD90, CD29 | – | [65] |
|
|
WT mice | CD29, CD90, CD44, CD105 | CD34, vWF | [66] |
|
|
C57/B16 | – | CD90, CD105, CD29, and CD133 | [67] |
|
|
C57BL/KsJdb/m | CD29, CD44, CD90, CD34, CD45 | CD34, CD45 | [68] |
|
|
Swiss albino mice | CD44, CD29 | – | [69] |
Conclusion
In this review we observed that researchers have used various CD markers for the phenotypic characterization of ADSCs. The most common CD markers are summarized in this review as a comprehensive map of which of them are highly expressed in the flow cytometric analysis of rat and mouse ADSCs. Although the accomplishment of a systematic review is not possible due to the absence of data (e.g., size, age, sex of the animals) in the relevant articles, we believe that this review will constitute the inception for the establishment of minimal criteria for the characterization of non-human ADSCs via flow cytometry.